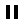Home »
Blog » Stem Cell
| Stem Cell, PRP, Acupuncture in Queens & Long Island, New York
Stem Cell | Stem Cell, PRP, Acupuncture in Queens & Long Island, New York
Tags: Stem Cell | Posted on: 08-Jan-2025 | No of views: 5079 Hip arthritis is a common yet often misunderstood condition that can significantly affect quality of life. Characterized by the deterioration of the cartilage in the hip joint, it leads to pain, stiffness, and difficulty in performing everyday activities. With advancements in medicine, particularly interventional pain management, there are effective ways to manage this condition and regain mobility and independence.
Read more
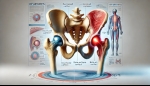
Tags: Stem Cell | Posted on: 30-Dec-2024 | No of views: 5373 What Are Elbow Sprains and Strains? An elbow sprain involves stretching or tearing the ligaments—the tough bands of tissue connecting bones—within the elbow joint. This typically occurs due to a sudden force, such as a fall onto an outstretched hand or a direct blow to the elbow. Conversely, an elbow strain refers to an injury of the muscles or tendons around the elbow, often resulting from overuse or excessive stretching.
Read more
Tags: Stem Cell | Posted on: 11-Aug-2023 | No of views: 5834 For people with a type of jaw joint disorder that results from loss of cartilage, the only treatments available address symptoms but do not repair the damaged tissue. Now, a new study of mice suggests stem cells already present in the jaw joint could be manipulated to repair it. The study, led by Columbia University Medical Center in New York, NY, is published in the journal Nature Communications. The authors describe how manipulating stem cells in the temporomandibular joint (TMJ) of mice with TMJ degeneration led the cells to repair cartilage in the joint.
Read more
Tags: Stem Cell | Posted on: 19-Jul-2023 | No of views: 6658 The field of regenerative medicine is rapidly expanding to help repair parts of your body that no longer function as well as they should due to injury or the natural aging process. The expert physicians provide patients with cutting-edge therapies of regenerative medicine. What is Regenerative Medicine? According to the National Institute for Health, regenerative medicine is the process of creating living, functional tissues to repair or replace tissue that no longer works as a result of disease, age, damage, or birth defects.
Read more
Tags: Stem Cell | Posted on: 05-Apr-2023 | No of views: 7698 About Stem Cell Therapy - Stem cells are found inside different types of tissue in the body. They are self-renewing and act as a repair system in the body. However, when the body gets injured, it may not be able to create enough stem cells to treat the appropriate area. This is why the interventional pain management specialist provides stem cell therapy in order to supply these cells to injured and painful joints, nerves, tendons, and muscles in order to promote healing and diminish any existing pain.
Read more
Tags: Stem Cell | Posted on: 17-Feb-2023 | No of views: 6714 Throughout your lifetime, specialized cells inside your body platelets and stem cells keep you healthy by continuously healing injuries and regenerating new cells to rebuild damaged tissues. Regenerative medicine taps into your body’s natural healing ability, retrieving these amazing cells and using them to treat your arthritic joints. The interventional pain management centers have extensive experience using regenerative medicine to ease your arthritis pain. Here, they explain the cells used, how they heal, and where they originate.
Read more
Tags: Stem Cell | Posted on: 29-Jul-2022 | No of views: 6520 There’s a simple reason why regenerative medicine can help you feel restored. It’s because regenerative treatments regrow and rebuild your tissues, so you are pretty literally revitalized. Of course, how that happens is a more complex story. The pain management centers specialize in regenerative medicine, so let’s talk about how it works, the conditions it can help heal, and the ways it will help you feel restored.
Read more
Tags: Stem Cell | Posted on: 13-May-2022 | No of views: 6659 The joint inflammation that defines arthritis can have a major impact on your mobility and quality of life. While there have been many advances in the field of joint replacement surgery, it’s still a last-resort treatment. The conventional wisdom in contemporary medicine leans toward exhausting all conservative treatments before recommending a surgical solution.
Read more
Tags: Stem Cell | Posted on: 20-Apr-2022 | No of views: 7119 Spinal pain can be debilitating. According to the American Chiropractic Association, it is estimated that 80% of people will experience back pain during their lifetime. The resulting stiffness from back pain can restrict one’s ability to move. Persistent pain may radiate to other parts of the body, including the chest, arms, and legs. As a result, walking properly and maintaining good posture can be difficult. For some patients, it may seem impossible to find a comfortable position since sitting, standing, and even sleeping may prove painful.
Read more
Tags: Stem Cell | Posted on: 02-Mar-2022 | No of views: 7400 Osteoarthritis (OA) remains to constitute a large burden to healthcare and negatively impact the quality of life; along with other conditions ranked as 11th highest contributors of global disability. The estimated prevalence of problematic hip/knee OA is approximately 242 million globally; 3.8% accounting for decreased quality of life (QoL) and societal burden.
Read more
Love this Post? Spread the World